As clinical trials become more complex and eligible patients become rarer, many studies fail to progress as planned. To address this, we formulate and execute novel “Solution Designs”—customized problem-solving strategies tailored to each trial—that lower the burden on medical institutions while making it easier for patients to participate.
Working alongside the sponsor, we identify the specific bottlenecks of each trial. We then drive the planning and execution of customized, novel solutions (“Qualitative Improvements”) to resolve these essential issues.
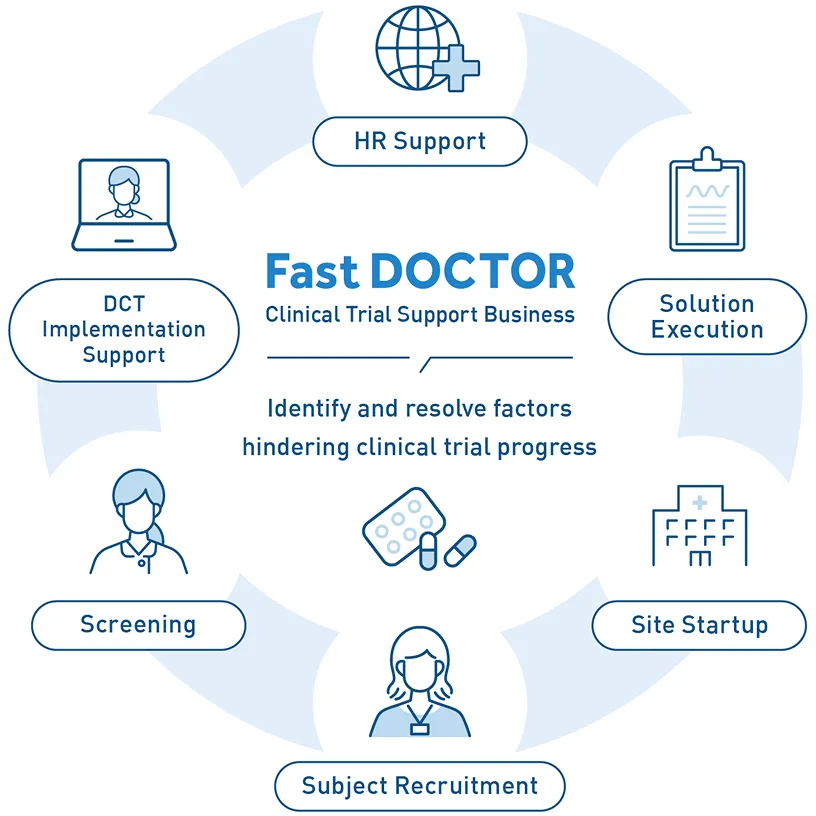
Issues to be tackled
Solutions to challenges
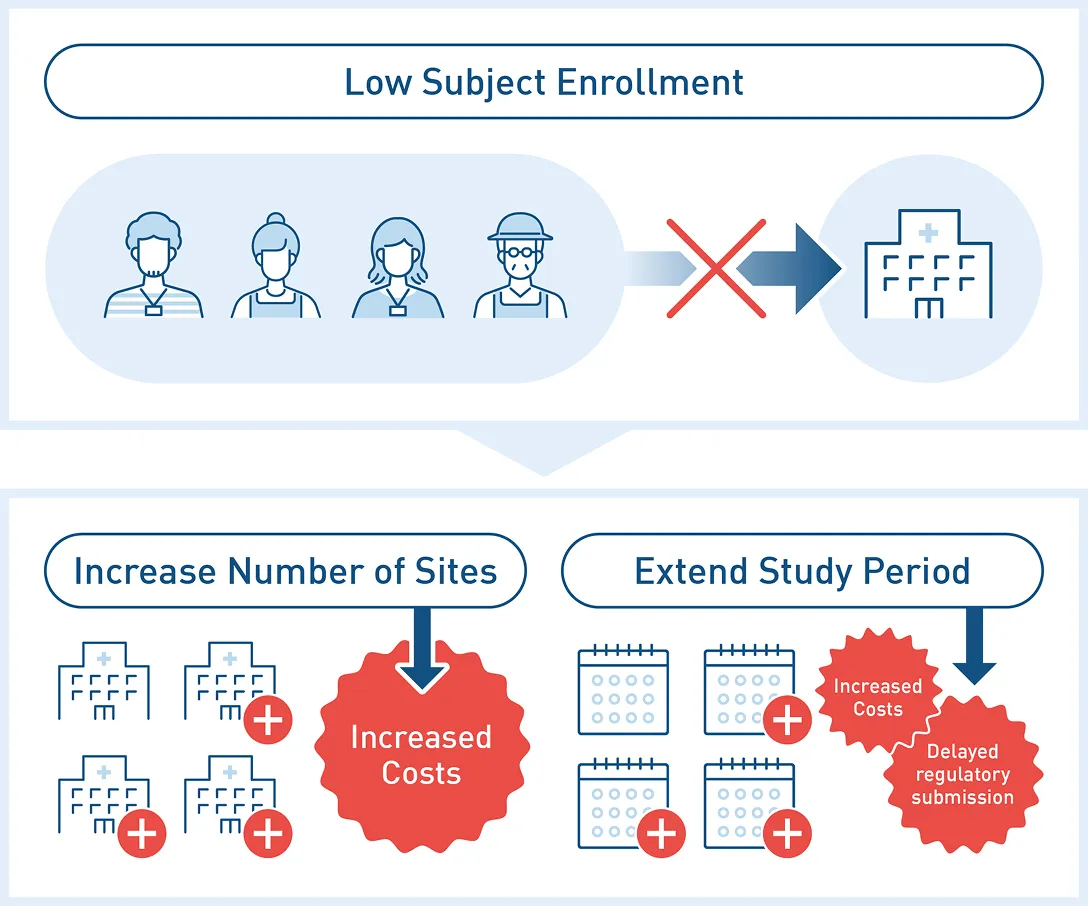
Japan has a large number of medical institutions compared to global standards, which tends to result in a lower number of subjects per facility.
Consequently, recruiting patients becomes difficult, impeding trial progress.
Attempting to accelerate trials by opening numerous sites causes costs to skyrocket.
Clinical trial costs in Japan are high compared to other nations.
This is considered a contributing factor to “Drug Loss”—a situation where drugs approved overseas remain unapproved in Japan.
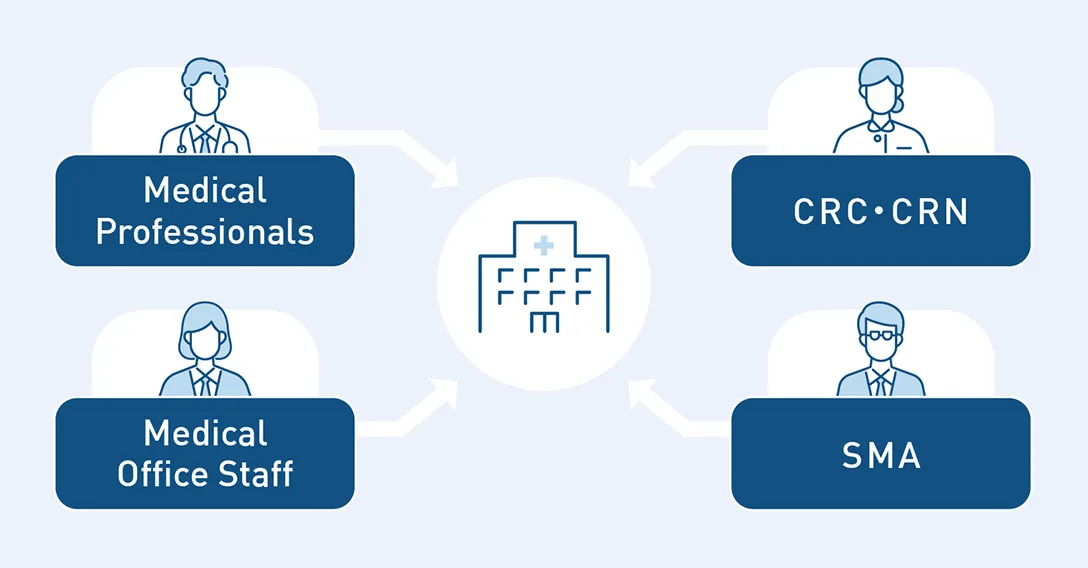
While Japan boasts a large number of medical institutions, the number of medical staff per facility is relatively low, leaving doctors overwhelmed by daily clinical practice.
As a result, they cannot allocate sufficient resources to clinical trials—another key reason for trial delays.
Furthermore, regarding the patients themselves, lifestyle diversification has made visiting medical institutions during weekday business hours a significant burden, creating a barrier to participation.
To resolve these complex, intertwined issues, it is essential to identify the root causes specific to each trial, design highly customized solutions, and build a viable execution structure.
There is a pressing need to create a new model for clinical trials.

Regarding subject recruitment, we utilize our network of partner medical institutions to refer patients to trial sites through a “Doctor-to-Doctor” referral scheme.
We have built a referral structure that guarantees both “Quality” and “Speed” by conducting screening based on high-quality, medical-record-driven data and providing timely trial information to potential subjects through a communication mix. For medical institutions outside our partnership network, we can also build referral schemes from scratch to meet client needs.

For facilities that have potential subjects but cannot adequately operate clinical trials due to busy daily practices, we provide HR support to resolve resource shortages.
Additionally, depending on the trial protocol, we provide hands-on educational support to help build a structure capable of conducting the trial.
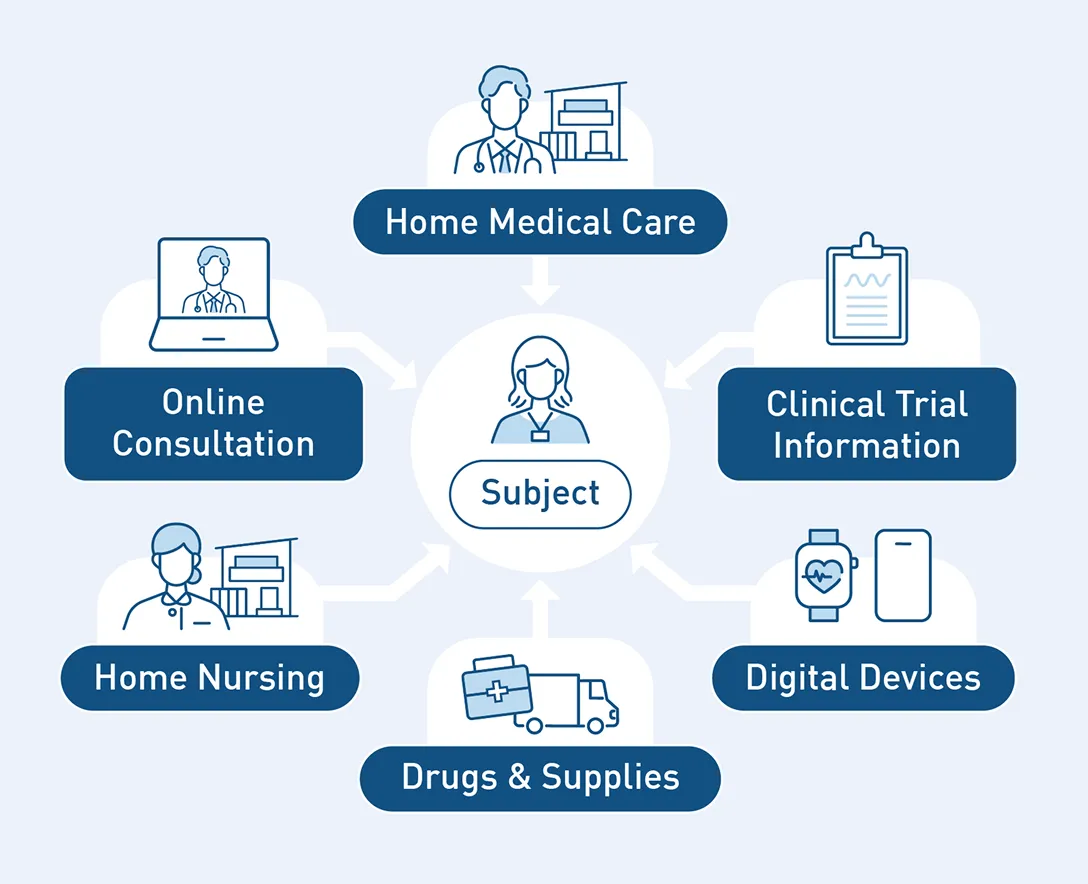
As a new form of clinical trial, we support the implementation of DCTs utilizing home visits and online medical care, leveraging Fast Doctor’s existing business expertise.
This contributes to lowering participation hurdles for pediatric patients, the elderly, and those with diseases that make hospital visits burdensome.
Clients often consult us because they are unsure where patients perceive barriers to participation.
We identify patients with attributes similar to the target profile, conduct interviews, and collect raw “Patient Voices”. Based on survey results, we conduct in-depth interviews to extract “Patient Insights”
—the root causes hindering participation—which are then utilized in formulating Clinical Development Plans.

Traditionally, when trials stalled, “Quantitative Improvements” such as adding sites or extending trial periods were common measures.
We go beyond quantitative measures.
By identifying bottlenecks specific to each trial and planning and executing “Qualitative Improvements” to resolve these root causes together with our clients, we contribute to the successful advancement of the study.